 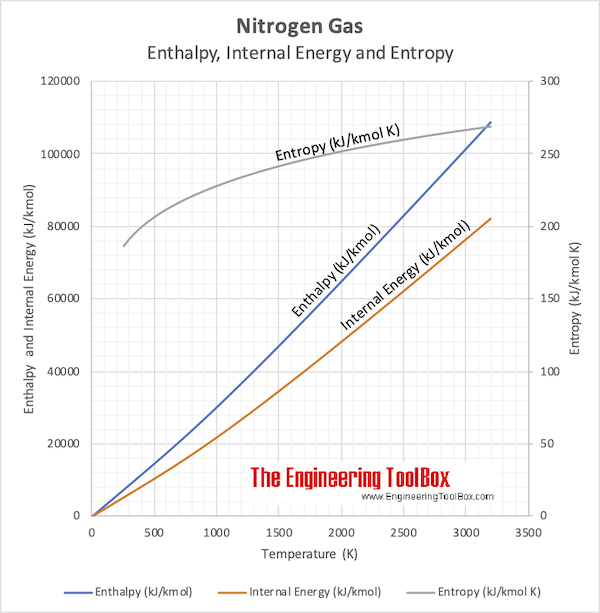
And, again, this spontaneous process is also characterized by an increase in system entropy. This supports the common observation that placing hot and cold objects in contact results in spontaneous heat flow that ultimately equalizes the objects’ temperatures. As for the previous example of matter dispersal, extrapolating this treatment to macroscopic collections of particles dramatically increases the probability of the uniform distribution relative to the other distributions. 
In 1865, Clausius named this property entropy ( S) and defined its change for any process as the following: Similar to other thermodynamic properties, this new quantity is a state function, and so its change depends only upon the initial and final states of a system. 
(a) Nicholas Léonard Sadi Carnot’s research into steam-powered machinery and (b) Rudolf Clausius’s later study of those findings led to groundbreaking discoveries about spontaneous heat flow processes. Note that the idea of a reversible process is a formalism required to support the development of various thermodynamic concepts no real processes are truly reversible, rather they are classified as irreversible. The term reversible process refers to a process that takes place at such a slow rate that it is always at equilibrium and its direction can be changed (it can be “reversed”) by an infinitesimally small change is some condition. This new property was expressed as the ratio of the reversible heat ( q rev) and the kelvin temperature ( T). In a later review of Carnot’s findings, Rudolf Clausius introduced a new thermodynamic property that relates the spontaneous heat flow accompanying a process to the temperature at which the process takes place. In 1824, at the age of 28, Nicolas Léonard Sadi Carnot ( Figure 1) published the results of an extensive study regarding the efficiency of steam heat engines.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
